US IVD Contract Manufacturing Market Growth and Industry Analysis By 2031
The United States diagnostic ecosystem is undergoing rapid transformation as healthcare providers and diagnostic companies increasingly rely on outsourced production models. Rising demand for advanced testing solutions, combined with the need for cost efficiency and faster product development cycles, is pushing manufacturers to collaborate with specialized contract service providers. This shift is strengthening the overall diagnostic value chain and improving access to high quality testing solutions across healthcare facilities.
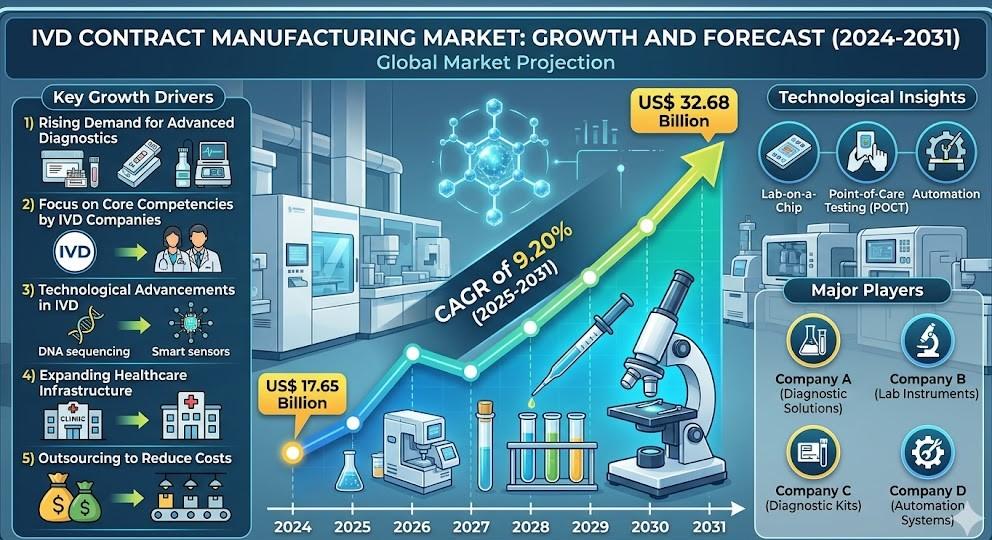
The US IVD Contract Manufacturing Market is experiencing strong expansion, supported by increasing adoption of molecular diagnostics, immunoassay platforms, and automated laboratory technologies. The market is benefiting from rising chronic disease prevalence, growing infectious disease testing requirements, and continuous innovation in diagnostic technologies. The segment is also gaining traction as companies focus on outsourcing manufacturing operations to improve scalability and maintain compliance with strict regulatory frameworks.
Expansion of Outsourcing in Diagnostic Manufacturing
One of the most significant trends shaping the US diagnostic production landscape is the growing preference for outsourcing manufacturing activities. Diagnostic companies are increasingly shifting away from in-house production to focus on research, innovation, and commercialization strategies.
Contract manufacturers provide end-to-end solutions, including product development support, large-scale manufacturing, packaging, and regulatory compliance. This allows diagnostic firms to reduce operational costs while improving production efficiency. The complexity of modern diagnostic products, such as multiplex assays and molecular testing kits, has further increased reliance on specialized manufacturing partners.
Strict regulatory requirements imposed by authorities such as the FDA also encourage outsourcing, as contract manufacturers are better equipped to maintain compliance and ensure product quality consistency.
Technological Advancements Reshaping Production Capabilities
Technological innovation is playing a central role in transforming diagnostic manufacturing processes in the United States. Molecular diagnostics, immunoassays, clinical chemistry, hematology, microbiology, and urinalysis technologies are all witnessing continuous advancements that require highly specialized production environments.
Molecular diagnostics is emerging as a key growth area due to its applications in genetic testing, oncology, and infectious disease detection. Contract manufacturers are investing in advanced PCR systems, sequencing technologies, and microfluidic platforms to meet rising demand for precision testing.
Automation is also reshaping production facilities. Robotics, artificial intelligence based quality control systems, and digital monitoring tools are improving manufacturing accuracy, reducing human error, and increasing throughput. These technologies are enabling manufacturers to meet high volume production requirements while maintaining strict quality standards.
Download Sample PDF @ https://www.theinsightpartners.com/sample/TIPRE00037805
Market Segmentation and Structural Overview
The US IVD contract manufacturing landscape is segmented based on product type and technology, each contributing significantly to industry growth.
Product segmentation includes devices, reagents, assay kits, and consumables. Among these, reagents and consumables dominate due to their recurring usage in diagnostic testing workflows. Devices also play an important role, especially in point-of-care and laboratory based diagnostic systems.
Technology segmentation includes:
- Immunoassay diagnostics
- Molecular diagnostics
- Clinical chemistry
- Hematology
- Microbiology
- Urinalysis
Immunoassay remains widely adopted due to its versatility and cost effectiveness. Molecular diagnostics is gaining momentum as healthcare shifts toward personalized medicine and genetic testing solutions. Other segments such as microbiology and hematology continue to support routine clinical diagnostics across hospitals and laboratories.
Key Growth Drivers Supporting Industry Expansion
Several structural factors are driving growth in the US diagnostic manufacturing outsourcing ecosystem. Increasing prevalence of chronic diseases such as diabetes, cardiovascular disorders, and cancer is significantly boosting diagnostic testing volumes.
Strategic collaborations between diagnostic companies and contract manufacturers are also accelerating innovation. These partnerships allow companies to leverage manufacturing expertise while focusing on product development and market expansion.
Another major driver is the increasing emphasis on quality assurance and regulatory compliance. Contract manufacturers with certified facilities and strong regulatory expertise are preferred due to their ability to ensure consistent product performance and global market readiness.
Rising demand for rapid and accurate diagnostic solutions, especially in infectious disease management, is further strengthening the need for scalable manufacturing infrastructure.
Competitive Landscape and Leading Companies
The competitive environment in the United States diagnostic manufacturing outsourcing sector is highly consolidated, with several global leaders and specialized firms operating across the value chain.
Key players include:
- Thermo Fisher Scientific
- Abbott Laboratories
- Roche Diagnostics
- Danaher Corporation
- Becton Dickinson and Company
- Siemens Healthineers
- bioMérieux
- Hologic
- PerkinElmer
- Ortho Clinical Diagnostics
These companies are actively investing in automation, expanding production capacities, and forming strategic partnerships to strengthen their position in the diagnostic manufacturing ecosystem. Continuous innovation and regulatory compliance remain key competitive differentiators.
Challenges Affecting Market Development
Despite strong growth potential, the industry faces several challenges. Maintaining consistent quality across large-scale production remains a critical concern, especially for complex diagnostic assays.
Supply chain disruptions and raw material shortages can impact production timelines and increase operational costs. Additionally, compliance with stringent regulatory requirements demands continuous investment in quality systems and certification processes.
Intellectual property protection and data security concerns in outsourced manufacturing environments also present ongoing challenges for both manufacturers and diagnostic companies.
Future Outlook
The future of the US diagnostic manufacturing outsourcing sector is expected to be shaped by continued technological advancement, increasing automation, and rising demand for personalized healthcare solutions. Molecular diagnostics and digital integration will remain key growth drivers, while artificial intelligence will further enhance production efficiency and quality control.
As healthcare systems continue to prioritize early disease detection and precision medicine, the role of contract manufacturers will become even more critical. Companies that invest in advanced technologies, regulatory excellence, and strategic collaborations will be well positioned to capture long term growth opportunities in this evolving ecosystem.
Related Reports @
Osteocalcin Assay Market Growth, Size, Share, Trends, Key Players and Forecast by 2034
Endotoxin Detection Service Market Outlook, Geography, and Dynamics by 2034
About Us -
The Insight Partners provides comprehensive syndicated and tailored market research services in the healthcare, technology, and industrial domains. Renowned for delivering strategic intelligence and practical insights, the firm empowers businesses to remain competitive in ever-evolving global markets.
Contact Us:
Ankit Mathur | The Insight Partners
E-mail: ankit.mathur@theinsightpartners.com
Phone: +1-646-491-9876
Also Available in : Korean| German| Japanese| French| Chinese| Italian| Spanish